Unlock your genetic potential or gift the power of personalized health. Browse our Supersaver Packs now.
The feeling of a good night’s sleep is second to none. Yet, for one reason or another, you could end up sleep-deprived on a given day. If this adds up, it could spell trouble for your organs and, most importantly, your heart. A recent study presented by China’s State Key Laboratory of Infectious Disease found an interesting link between weekend naps and your cardiovascular health. Just how potent are those weekend naps? Read on to find out.
Sleep is the downtime your body needs to set itself right each night.
While you sleep, you recharge, reinforce your body systems, regulate your metabolism, and prepare for the next day.
This study defines sleep deprivation as under 7 hours of sleep per night.
Prolonged sleep deprivation can lead to problems with metabolism and the working of your organs.
Sleep-deprived people fall into certain patterns of behavior, including:
All of these are risk factors for heart disease.
That’s why striving for a good night’s sleep is essential.
The researchers aimed to find a link between irregular sleep patterns and cardiovascular disease (CVD) risk.
They defined two essential terms for the study:
They studied data from 90,903 people from the UK Biobank.
The UK Biobank comprises 500,000 participants’ physical and medical assessment data, curated from 2006 to 2010.
Researchers divided the study group into cohorts, depending on how much compensatory sleep they had gotten.
They used accelerometers to study the sleep patterns of the subjects.
Finally, around one-fifth of the participants (21.8%) were classified as sleep-deprived.
Initially, all participants were free from any kind of CVD.
The researchers observed them until their first major CVD event.
Limited ethnic representation: Most of the UK Biobank comprises white British people of higher socioeconomic classes. This limited the generalization of the study’s results.
However, even after adjusting for several factors, the association between sleep quality and the risk of CVD remained strong.
The researchers advocated for promoting regular sleep duration to reduce your risk of CVD.
This study recommends sleeping more to cut down the risk of heart problems.
Additionally, there are more things you can do to keep your heart healthy.
Some of these include:

What Does It Mean When The Ring Finger Is Longer Than The Index Finger?

Paper Straws: Not A Safer Option For Your Health Or The Environment

Do Tattoos Cause Cancer Like Lymphoma?
Put your hand out in front of you. What do you see? Is your index finger longer or shorter than your ring finger? For centuries, depending on the length of their digits, people have classified themselves into archetypes with defined personality traits. Does this classification hold up in today's day and age, where research and science are the final authority? Could the size of your ring finger indicate your tendency for traits like risk-taking or assertiveness? Read on to find out what science says.
Your finger length ratio is the length of your index finger compared to the length of your ring finger.
Also called the 2D:4D ratio, this measure has become controversial in scientific research.
While some research suggests the ratio is a putative marker for testosterone exposure during pregnancy, others argue that its statistics are flawed and can’t be reproduced validly.
Certain conditions in the womb influence the 2D:4D ratio in the fetus.
Here’s what you need to know:
However, critics argue that this measure is simplistic and not statistically valid.
Some even contend that this area of study has irreproducible findings.
If your ring finger is longer than the index finger, you have a low 2D:4D ratio.
Some studies say this may be due to a greater prenatal exposure to testosterone.
Consequently, this could also impart further “masculine” traits to people with longer ring fingers and more “feminine” traits to people with longer index fingers.
A longer ring finger may also be associated with better athletic performance, cognition, and risk-taking behavior.
We’ll learn more about this in a while.
Research suggests that genetic influences account for about 60-70% of the variability in the finger length ratio.
The SMOC1 and LIN28B genes essential for early limb development may be responsible.
A study conducted by Cambridge University found a variant in the SMOC1 gene.
One form of this variant was associated with a longer ring finger, and the other with a longer index finger.
Some reports suggest the influence of testosterone and estrogen levels in controlling the activity of this gene.
Two studies presented at the Alzheimer's Association International Conference explored the link between digit length and cognitive impairment or dementia in older adults.
The study reported that females with longer index fingers scored higher on cognitive tests at younger ages, consistent with better verbal abilities.
Older females with longer ring fingers performed better than those with longer index fingers.
However, the study found no statistically significant differences for men.
The results suggest that prenatal testosterone exposure may help preserve cognition in older women, potentially reducing their vulnerability to Alzheimer's disease.
However, there are several limitations of this study:
Self-reporting biases, potential confounding variables, and the lack of longitudinal data further limit the robustness of the conclusions.
Though not fully proven, some experts believe that the 2D:4D ratio (index finger to ring finger) can be used to predict athletic prowess.
A study has reported that teenage boys with a longer ring finger (lower 2D:4D ratio) have better hand grip strength.
A finger length ratio expert conducted a study in 2024 examining the relationship between finger length and oxygen consumption (an indicator of athletic performance).
He found that those with longer a ring finger metabolized oxygen more efficiently in a cardio test.
A study investigated finger length ratios and their link to “masculinity” and “femininity” in nearly 1000 women.
The analysis revealed that women with shorter index fingers were more likely to describe themselves as ‘assertive and competitive.’
However, this study had significant limitations:
A study used data from an extensive population survey to decode the association between finger length ratio and risk behavior.
Here's what they found:
The researchers found this pattern in both men and women and for both hands.
However, the connection isn't solid - it's just a small tendency, not a rule that applies to everyone.
The size of men's hands has been a subject of scientific inquiry and widespread speculation.
While some claims about hand size are based on myths or unfounded assumptions, researchers have studied a few aspects scientifically.
Here's a comprehensive overview:
| Trait | Correlation |
| Hand Size & Height | Taller individuals generally have larger hands |
| Hand Size & Grip Strength | Larger hands are often correlated with better grip strength. However, muscle mass and training can also make a difference. |
| Hand Size & Testosterone Levels | Some studies say more testosterone equals larger hands. This relationship needs to be studied further. |
While studies have shown that finger ratio can affect other traits, there are many myths surrounding this topic.
Some of these misconceptions to watch out for include:

The Link Between Vitamin D And Raynaud's Disease
5 Things You Should Know Before Buying Xcode Life
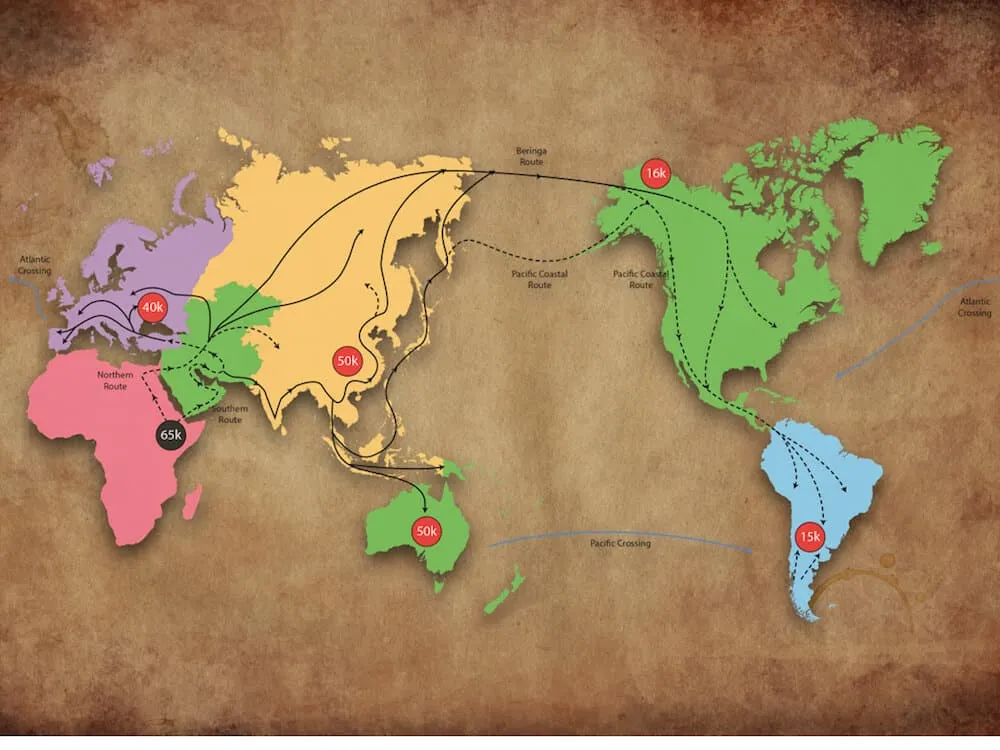
How Far Back Do Ancestry DNA Tests Go?
https://udel.edu/~mcdonald/mythfingerlength.html
https://www.23andme.com/topics/traits/finger-length-ratio
https://www.lesmills.com/us/fit-planet/health/finger-length
https://www.livescience.com/49883-finger-length-in-men.html
https://www.sciencedirect.com/science/article/abs/pii/S0191886914006400
Your DNA is the most personal, sensitive piece of information that is uniquely yours. When you entrust it to a company, you should be certain they will use it responsibly. Xcode has always stood out as a sincere, honest service that prioritizes user privacy above all else. Privacy is an integral part of our DNA, and we continue to strive to provide a secure platform for you, the user. Our privacy policy remains clear-cut and concise, reminding you that at Xcode Life, your privacy comes first.
DNA data privacy involves protecting a person's genetic information.
Storage and repurposing of a user’s DNA data must be done transparently.
Sharing DNA with third parties must require the user’s well-informed consent.
Federal laws, such as the Common Rule and the Health Insurance Portability and Accountability Act (HIPAA), aim to protect consumer privacy while encouraging scientific progress.
When you choose Xcode, your focus should be solely on learning more about your health.
You will remain in charge of your DNA data from the moment you upload it to our site.
Here’s how you can control your data through Xcode.
Your DNA data deserves nothing less than the best security measures.
We review and update our privacy measures regularly.
Your uploaded data is encrypted, anonymized, and stored safely within our secure databases.
Once you upload your data, it will stay on our secure server.
Your data isn’t shared with any third-party tools, even anonymously.
No insurance companies or employers will have access to your data.
We intend to empower you to take charge of your health and have no intention of selling your data for commercial gain.
Only a handpicked set of trained employees have access to your DNA data.
They have undergone training to handle your data securely.
They are bound by confidentiality protocols, meaning nobody else at Xcode can access your data.
We access your data for only two things:
If you do not wish for your data to remain on our server once you receive the report, you can delete it through your account by contacting us, as mentioned previously.
With Xcode Life, you can be confident that we handle your data in the most secure way possible.
You can focus on gaining actionable insights to build a healthier future.
We hope that we’ve addressed your privacy concerns in this post.
If you have any other questions, please do not hesitate to contact us.
We will always do our best to provide you with the information you require.

5 Things You Should Know Before Buying Xcode Life
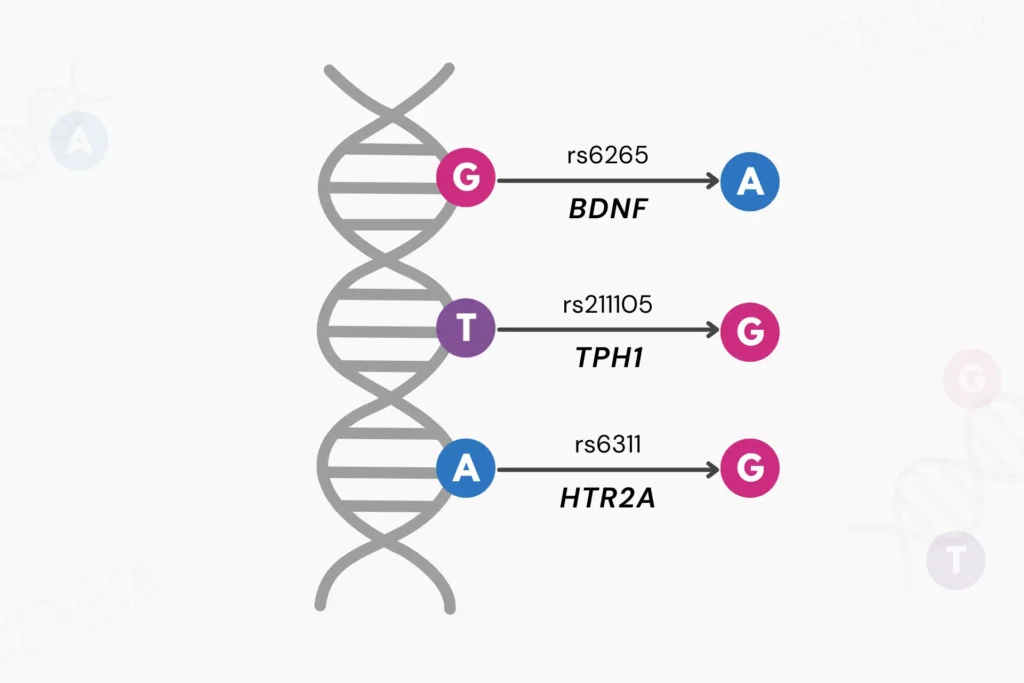
What Are RSIDs?

How Genes Influence Your Risk For Lactose Intolerance
Newborn babies can be prone to bleeding disorders, and one standard medical procedure to combat this involves administering a vitamin K shot at birth. Vitamin K is known to help promote clotting and blood health by nourishing the liver. However, there are pros and cons to consider when deciding whether or not to opt for a vitamin K shot for your newborn. This article will explore the benefits and risks of administering a vitamin K shot at birth.
Vitamin K is a fat-soluble vitamin found in two forms in your food.
It helps make the proteins needed for blood clotting
Vitamin K deficiency can lead to low levels of prothrombin in the blood.
Prothrombin is a blood clotting protein whose deficiency can lead to hemorrhage (internal bleeding) and blood loss.
Vitamin K deficiency is very rare in adults.
It occurs in individuals with malabsorption issues or those taking medications that interfere with vitamin K absorption.
According to the National Institute of Health (NIH), infants between 0 and 6 months need two micrograms of vitamin K daily to maintain adequate nutritional levels.
This is because every newborn child has low vitamin K levels in their blood.
Two main reasons for this are:
Low vitamin K levels in infants put them at a higher risk for developing a condition called Vitamin K Deficiency Bleeding (VKDB) in the first six months of their life.
This is why every infant needs a vitamin K shot at birth.
Since 1961, the American Academy of Pediatrics has recommended a vitamin K shot at birth to prevent the risk of VKDB.
VKDB causes life-threatening internal bleeding, which can occur within 6 months of birth.
According to experts, one out of five babies with VKDB die because of the condition.
Two out of every five survivors end up with long-term neurological damage.
They may also have motor skill defects, cognitive issues, and organ failures.
VKDB may also lead to motor skill defects, cognitive issues, and organ failures in surviving children.
Early VKDB occurs within 24 hours of birth.
The baby could be born with an existing bleeding disorder, which may worsen due to low vitamin K levels.
The use of certain maternal medications also increases the risk of early VKDB.
Classic VKDB occurs between 24 hours and one week of birth.
It’s caused by a natural decrease in vitamin K levels, which breast milk cannot adequately replenish.
Late VKDB occurs between one and 24 weeks (6 months) of birth.
It could result from the baby's existing liver problems, which make the liver unable to produce clotting factors.
All three cases of VKDB cause bleeding in the gut or brain and can turn severe quickly.
Doctors usually administer Vitamin K as a single intramuscular shot or an oral dose right after birth.
The injection contains vitamin K and certain safe additives that maintain the vitamin's pH levels and improve its absorption rate.
A single shot of vitamin K within 6 hours of birth can greatly reduce the risk of VKDB.
The dose is generally 1mg, but babies weighing under 1500g at birth get a 0.5mg dose of vitamin K.
| Vitamin K Injection | Vitamin K Tablets |
| One-time procedure | Three doses over 8 weeks |
| Delivered intra-muscularly | Delivered orally |
| Absorbed quickly and efficiently | Absorbed slowly and partially |
| Recommended by the CDC | Not recommended by the CDC |
In 2019, a study surveyed parents who were against vitamin K shots and noted a few reasons:
A growing number of people oppose vitamin K shots at birth.
It’s essential to clearly explain the pros and cons to help them make an informed decision.
| Pros of Vitamin K Shot At Birth | Cons Of Vitamin K Shot At Birth |
| Prevents VKDB, a rare but severe blood clotting problem in infants | Rare instances of infection, bleeding, or bruising at the site of injection |
| Easy administration; intramuscular(recommended) or oral | Preservatives like benzyl alcohol can be toxic in large amounts for very premature babies |
| One of the safest substances to administer |
Some parents wanted to wait until their infant showed signs of vitamin K deficiency.
However, visible signs of VKDB appear only after significant internal bleeding since it starts affecting the liver right from birth.
If the condition grows severe, there is little that doctors can do.
In most countries, hospitals offer vitamin K shots at birth completely free of cost.
Yes, VKDB incidences in the USA are extremely low.
It’s only because nearly all newborns take the vitamin K shot.
VKDB is more common in regions where vitamin K shots aren’t readily available.
Yes. Vitamin K isn’t properly delivered to the fetus through the placenta and won’t be adequately delivered through breast milk.
Every newborn needs a vitamin K shot to prevent internal bleeding due to VKDB.

The Link Between Vitamin D And Raynaud Disease

How Long Do Vitamins Take To Work?

5 Things You Should Know Before Buying Xcode Life
With over 6 million people affected by GERD globally in 2019—an alarming 77.19% increase from 3.4 million in 1990—the prevalence of this troubling condition is rising rapidly. GERD, characterized by the reverse flow of stomach acid into the esophagus, is a widespread health concern with significant genetic influences. Research suggests that hereditary factors play a major role in an individual's risk of developing GERD. Knowing how genetics play a role is vital for managing and potentially avoiding this chronic disease. Keep reading to discover how your genetics might impact your risk of GERD and how this knowledge can inform your health choices.
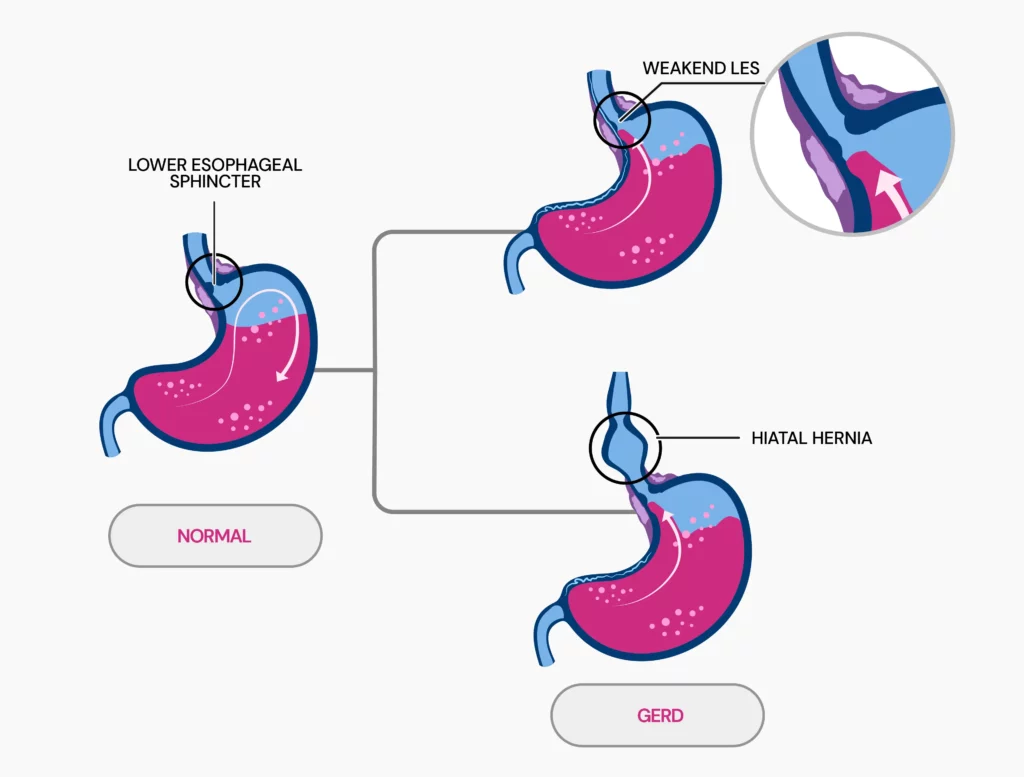
Gastroesophageal reflux disease (GERD) occurs when stomach acid flows back into the esophagus.
This occurs due to a problem in the lower esophageal sphincter (LES), a muscle ring that separates the esophagus from the stomach.
Normally, this ring opens to allow food into the stomach and then closes to prevent the stomach's contents from flowing back up.
In GERD, the LES is either weak or relaxes when it shouldn't, allowing acid to move up into the esophagus.
Fundamental causes of GERD:
The most common cause of GERD is a weak or relaxed LES.
When the LES becomes weak or doesn't function properly, stomach acid is permitted to flow back into the esophagus, resulting in the symptoms associated with GERD.
Yes, GERD can be hereditary. Research reveals that genetic factors play a significant role, with a heritability rate of about 31%.
Certain genetic markers, like specific single-nucleotide polymorphisms (SNPs), are linked to a greater risk of developing GERD.
Several genes are linked to GERD. Key genes include:
Genes like COX-2, IL-10, GSTP1, XRCC1, hMLH1, and A61G are also associated with increased GERD risk due to their roles in inflammation, DNA repair, and growth factors.
Yes, acid reflux, also known as GERD, can run in families. A twin study provides strong evidence for a genetic component in GERD.
The study examined 1960 twin pairs, including both identical (monozygotic) and fraternal (dizygotic) twins.
Identical twins, sharing the same set of genes, were more likely to have GERD than fraternal twins, who share only half their genes.
The study indicated that monozygotic twins had a GERD symptom rate of 42%, compared to 26% for dizygotic twins. This suggests a significant genetic influence on GERD.
The study’s heritability estimates indicate that around 43% of the variance in GERD risk is due to genetic factors.
This suggests that having family members with GERD increases your chance of getting it, pointing to a strong genetic influence.
Several conditions and lifestyle factors can increase the risk of GERD. Key contributors include:
Underlying conditions:
Lifestyle factors:
Yes, GERD is often a lifelong condition. It’s a chronic problem, which means that even if symptoms improve with treatment, they often come back when treatment stops.
People with severe GERD, like those with erosive esophagitis, usually need long-term treatment to keep the symptoms under control and prevent complications.
Studies have shown that after stopping treatment, up to 90% of people with healed erosive esophagitis see their symptoms return within 6 to 12 months.
This shows that GERD doesn’t go away easily and often requires ongoing management.
Lifestyle changes often serve as the first line of treatment for GERD. If these adjustments are insufficient, further medical intervention may be necessary.
Over-the-counter options:
Prescription treatments:
Surgical options:
For obesity-related GERD, weight-loss surgery may be recommended.
Note: Always seek advice from a healthcare professional before starting any treatment or medication for GERD to ensure it's the right approach for your condition.
Dr. Yalini Vigneswaran, MD, MS, a gastrointestinal surgeon at the University of Chicago Medicine, offers expert advice on managing and preventing GERD, including:

Is Lupus Hereditary? An Overview of Genetic Factors

How It Works: Are Freckles Genetic?

Is Handedness Genetic? What Science Tells Us
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9122392
https://www.webmd.com/heartburn-gerd/reflux-disease-gerd-1
https://www.medicalnewstoday.com/articles/14085
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6107529
https://gut.bmj.com/content/52/8/1085.full
https://my.clevelandclinic.org/health/diseases/17019-acid-reflux-gerd
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6140167
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1936305
https://www.mayoclinic.org/diseases-conditions/gerd/diagnosis-treatment/drc-20361959
There’s a reason doctors insist you take your vitamins. The tingling sensation that Raynaud’s disease causes in your fingers could lead to more than mild irritation. Raynaud's disease occurs when blood stops flowing to your hands and fingers in cold or stressful situations. The most mundane of tasks can become a challenge during a flare-up. While the exact cause of Raynaud's is still being pieced together, research suggests that Vitamin D plays a significant role in maintaining healthy blood circulation. This article will explore the intriguing connection between vitamin D and Raynaud disease.
Raynaud’s disease causes a reduction in blood flow to your extremities (hands and feet).
It affects women more often than men.
It is usually triggered by stress or cold temperatures.
Reduced blood flow to the extremities can make them look pale, blue, or red.
A tingling sensation accompanies these color changes.
In Raynaud's disease, blood vessels overreact to cold or stress and undergo vasoconstriction.
It is usually caused due to the deficiency of certain minerals (like magnesium) in the body.
Structural abnormalities in the blood vessels can also cause Raynaud's disease.
Raynaud's phenomenon can be of two types: primary and secondary.
| Primary Raynaud’s | Secondary Raynaud’s |
| Easier to manage | Harder to manage |
| More common | Less common |
| No permanent damage | Causes tissue damage, gangrene, and sometimes loss of fingers or toes |
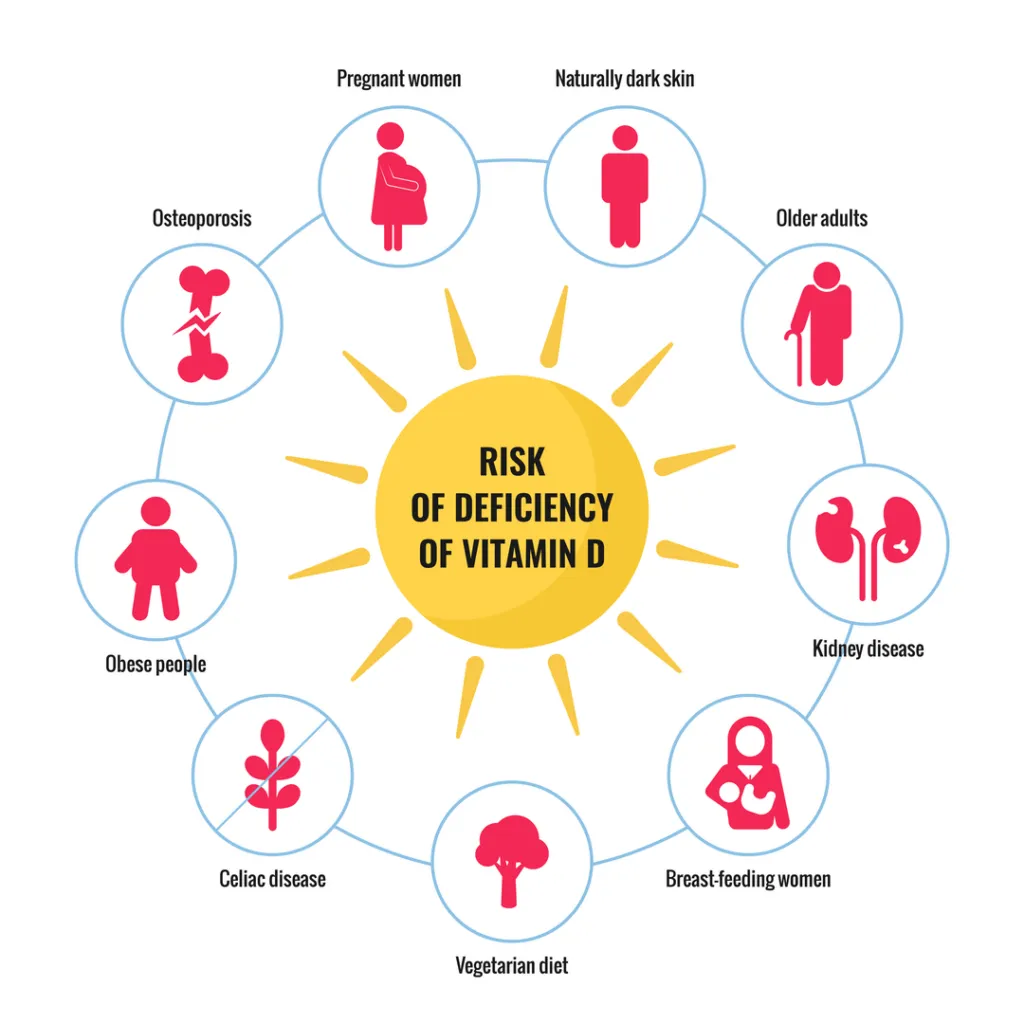
Some research shows that having adequate vitamin D levels in the body might help with Raynaud's.
Smooth muscles line blood vessels.
Vitamin D relaxes these smooth muscles, thus preventing vasoconstriction.
Some studies show that vitamin D is an anticoagulant.
It has a blood-thinning effect.
Thus, vitamins could help manage Raynaud's.
In a study, 42 people with vitamin D deficiency were given oral supplements of vitamin D3.
The subjects reported that the severity of Raynaud's significantly decreased after 12 weeks.
However, more research is required to back this claim.
So far, studies say that Vitamin D might help ease Raynaud's symptoms.
However, more research is required to back this claim.
People suffering from Raynaud's disease should meet their vitamin D3 levels.
It can be by taking supplements after consulting with your doctor.
You could add fatty fish like salmon and fish liver oil to your diet for adequate vitamin D3.
Ideally, your vitamin D3 levels should be between 20 and 50 ng/ml.
It can be taken either as a supplement or through the diet as fish like salmon.
When exposed to the cold, Raynaud’s disease causes numbing in the hands and feet.
Primary Raynaud’s causes some numbness and is not long-lasting.
Secondary Raynaud’s can be more severe and might require medical intervention.
Raynaud’s can result from environmental changes or underlying diseases like lupus, rheumatoid arthritis, or scleroderma.
Vitamin D can potentially improve Raynaud’s symptoms by promoting circulation.
Ginkgo has been shown to dilate blood vessels and ease circulation, easing pain in patients with Raynaud’s.
Consult your doctor before taking supplementation for your condition.
Naturally, colder foods can cause a flare-up of Raynaud’s.
Caffeine causes your blood vessels to narrow, so it may be a trigger as well.
People have reported going months without a flare-up or severe symptoms.
In some cases, the condition has gone away altogether.

How Long Does It Take For Vitamins To Work?

Vitamin K Shot At Birth: Pros And Cons
Top 7 Vitamins and Nutrients For Clearing Brain Fog